| 中文名称 |
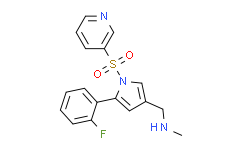
Vonoprazan
|
| 中文别名 |
5-(2-氟苯基)-N-甲基-1-(3-吡啶基磺酰基)-1H-吡咯-3-甲胺;氟呐普拉赞;沃诺拉赞
|
| 英文名称 |
Vonoprazan
|
| 英文别名 |
Vonoprazan;TAK-438 free base;TAK-438;TAK438 free base;TAK-438 (free base);1R5L3J156G;1-[5-(2-fluorophenyl)-1-pyridin-3-ylsulfonyl-pyrrol-3-yl]-~{N}-methyl-methanamine;1-(5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl)-N-methylmethanamine;Vonoprazan [INN];1-[5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1h-pyrrol-3-yl]-n-methylmethanamine;BCP13240;1-[5-(2-fluorophenyl)-1-pyridin-3-ylsulfonylpyrrol-3-yl]-
|
| Cas No. |
881681-00-1
|
| 分子式 |
C17H16FN3O2S
|
| 分子量 |
345.39
|
| 包装储存 |
Powder -20°C 3 years;4°C 2 years
|
| 详情描述 |
Vonoprazan (TAK-438 free base) 是一种质子泵抑制剂 (PPI),是高效的,具有口服活性的钾竞争性酸阻断剂 (potassium-competitive acid blocker, P-CAB),具有抗分泌活性。在 pH 为 6.5 时,Vonoprazan 抑制猪胃微粒体中的 H+,K+-ATPase 酶活性,IC50 值为 19 nM。Vonoprazan 被开发用于研究胃酸相关疾病,如胃食管反流病和消化性溃疡。Vonoprazan 可用于根除幽门螺杆菌。
|
| 产品详情 |
Vonoprazan (TAK-438 free base) 是一种质子泵抑制剂 (PPI),是高效的,具有口服活性的钾竞争性酸阻断剂 (potassium-competitive acid blocker, P-CAB),具有抗分泌活性。在 pH 为 6.5 时,Vonoprazan 抑制猪胃微粒体中的 H+,K+-ATPase 酶活性,IC50 值为 19 nM。Vonoprazan 被开发用于研究胃酸相关疾病,如胃食管反流病和消化性溃疡。Vonoprazan 可用于根除幽门螺杆菌。
|
| 生物活性 |
Vonoprazan (TAK-438 free base), a proton pump inhibitor (PPI), is a potent and orally active potassium-competitive acid blocker (P-CAB), with antisecretory activity. Vonoprazan inhibits H,K-ATPase activity in porcine gastric microsomes with an IC 50 of 19 nM at pH 6.5. Vonoprazan is developed for the research of acid-related diseases, such as gastroesophageal reflux disease and peptic ulcer disease. Vonoprazan can be used for eradication of Helicobacter pylori.
|
| 性状 |
Solid
|
| IC50 & Target[1][2] |
IC50: 19 nM (porcine gastric H,K-ATPase, at pH 6.5)
|
| 体外研究(In Vitro) |
Vonoprazan (0.1 nM-10 μM; 30 minutes) exhibits porcine gastric H, K-ATPase activity in a concentration-dependent manner.Vonoprazan does not inhibit Na,K-ATPase activity, even at concentrations 500 times higher than their IC50 values against gastric H,K-ATPase activity. has not independently confirmed the accuracy of these methods. They are for reference only.
|
| 体内研究(In Vivo) |
Vonoprazan (1-4 mg/kg; p.o.) completely inhibits basal and 2-deoxy-D-glucose (200 mg/kg; s.c.)-stimulated gastric acid secretion at the 4 mg/kg dose in rats. has not independently confirmed the accuracy of these methods. They are for reference only.
Animal Model:
|
| 运输条件 |
Room temperature in continental US; may vary elsewhere.
|
| 储存方式 |
Powder -20°C 3 years;4°C 2 years
|
| ClinicalTrial |
|
| 参考文献 |
[1]. Arikawa Y, et al. Discovery of a novel pyrrole derivative 1-[5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine fumarate (TAK-438) as a potassium-competitive acid blocker (P-CAB). J Med Chem, 2012, 55(9), 4446-4456.[2]. Hori Y, et al. 1-[5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine monofumarate (TAK-438), a novel and potent potassium-competitive acid blocker for the treatment of acid-related diseases. J Pharmacol Exp Ther, 2010, 335(1),
|
| 溶解度数据 |
In Vitro: DMSO : 100 mg/mL (289.53 mM; Need ultrasonic)配制储备液
|
[1]. Arikawa Y, et al. Discovery of a novel pyrrole derivative 1-[5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine fumarate (TAK-438) as a potassium-competitive acid blocker (P-CAB). J Med Chem, 2012, 55(9), 4446-4456.[2]. Hori Y, et al. 1-[5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine monofumarate (TAK-438), a novel and potent potassium-competitive acid blocker for the treatment of acid-related diseases. J Pharmacol Exp Ther, 2010, 335(1),
1:一般建议:溶解度为Medlife测试数据,可能与文献描述存在差异。这是由于生产工艺和批次不同产生的正常现象。为了使其更好的溶解,请用37℃加热试管并在超声波水浴中震动片刻。不同批次产品溶解度各有差异,仅做参考,具体以实验方案为准。
2:储存条件:粉末-20°C一般情况可以保存3年,溶于溶剂-80°C一般情况可以保存1年。不同产品及不同批次产品可能存在差异,请细致阅读产品信息,并辅助参考相关文献描述。


 扫码关注公众号
扫码关注公众号